Kyo-diagnostics Co., Ltd.
Prducts/Service
Drug sensitivity testing using cancer stem cell spheroids and related applications.
Kyo-diagnostics maintain a collection of more than 300 types of patient-derived cancer stem cell spheroids. These cancer stem cell spheroids can be utilized not only for fundamental and translational cancer research, but also for the development of innovative next-generation anticancer therapeutics.
Drug Sensitivity Testing Using Cancer Stem Cell Spheroids
Drug Sensitivity Testing on Target Cancer Stem Cell Spheroids
(Some cell lines are linked to clinical data or have undergone genomic analysis, enabling testing with cells selected according to your objectives, such as drug resistance profiles, genetic mutations, and gene expression patterns.)
Drug Sensitivity Testing Using a Cancer Stem Cell Spheroid Panel
Drug Sensitivity Testing Using a Diverse Panel of Cancer Stem Cell Spheroids
(This assay enables evaluation of drug selectivity and efficacy across various cell types, allowing determination of which cells respond or fail to respond to a given agent. It also facilitates assessment of the breadth of cellular response, and can identify agents that are selectively effective only in cells within the panel, even when those agents have shown no efficacy elsewhere.)
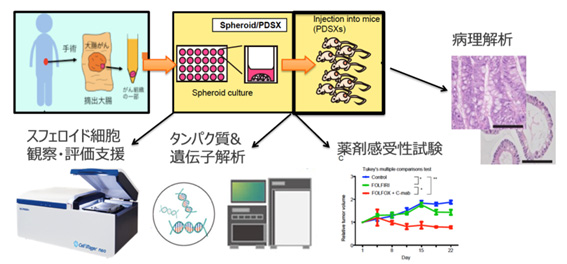
PDSX generation and drug sensitivity testing using cancer stem cell spheroids.
In vivo Testing Using a Mouse Xenograft Model (PDSX) Engrafted with Cancer Stem Cell Spheroids
(Faster model generation compared to PDX, with reduced variability, enabling reliable evaluation with smaller sample sizes.)
Genomic Mutation and Expression Analysis Before and After Drug Response
DNA/RNA Sequencing, mRNA Expression Analysis, ELISA, Western Blotting, and More
CTC (Circulating Tumor Cell) analysis and quantification.
- ・Non-invasive, Label-free Isolation of CTCs from Blood (e.g., without the use of antibodies)
- ・Expansion culture of circulating tumor cells (CTCs) and comprehensive analyses, including DNA/RNA sequencing, mRNA expression profiling, ELISA, and Western blotting.
Measurement of ctDNA (circulating tumor DNA).
- ・Isolation of circulating tumor DNA (ctDNA) from blood samples.
- ・Sequencing analysis of circulating tumor DNA (ctDNA).
Cancer Type
Colorectal, Small Intestine, Duodenum, Gastric, Lung, Pancreatic, Renal, Breast, Head and Neck, Prostate, Ovarian, Uterine, Brain, Rare Cancers
*Please contact us regarding your cancer type of interest.
Target Modalities
Small Molecules, Peptides, Nucleic Acids, Antibodies, ADCs (Antibody-Drug Conjugates)
Reserch
The incidence of colorectal cancer is estimated at approximately 158,000 cases domestically (2016) and around 1.8 million cases worldwide (2018). There is a growing demand for the development of simpler and more accurate diagnostic agents and methods for assessing drug responsiveness and prognosis in colorectal cancer.
In collaboration with the Graduate School of Medicine at Kyoto University, the company offers cancer diagnostics-related products and services contributing to colorectal cancer treatment, based on technologies developed through research on metastasis and invasion mechanisms in the Pharmacogenomics Unit, as well as individualized medicine research in gastrointestinal surgery.
Research Using PDSX Models Derived from Human Colorectal Cancer Stem Cell Spheroids
- ・Compared to conventional Patient-Derived Xenograft (PDX) models of colorectal cancer, we have demonstrated that the Patient-Derived Spheroid Xenograft (PDSX) model — in which gastrointestinal cancer cells cultured as spheroids are engrafted into mice — achieves a higher engraftment rate and shorter housing period.
- ・In the PDSX model, human-derived colorectal cancer stem cell spheroids, rather than established cell lines, are used. This enables the formation of tumor histology highly similar to that of primary colorectal cancer lesions (without stromal cells), and provides high-quality drug sensitivity responses.
References
Molecular Cancer Therapeutics, 2018 Oct;17(10):2187-2196. A Chemosensitivity Study of Colorectal Cancer Using Xenografts of Patient-Derived Tumor Initiating Cells. Maekawa H, et al.
Oncotarget. 2018 Apr 24;9(31):21950-21964. An improved method for culturing patient-derived colorectal cancer spheroids. Miyoshi H, et al
Conference presentation
- ・16th Annual Meeting of the Conference on Biomolecular Screenology: November 27–28, 2025, Tokyo (Tower Hall Funabori), Booth Exhibition
- ・38th Annual Meeting of the Japanese Society for Alternatives to Animal Experiments: November 1–3, 2025, Yokohama, Corporate Exhibition Booth
- ・84th Annual Meeting of the Japanese Cancer Association: September 25–27, 2025, Kanazawa, Corporate Exhibition Booth (within SCREEN Corporation’s Booth)
- ・29th Annual Meeting of the Japanese Association for Molecular Target Therapy of Cancer: July 2–4, 2025, Sapporo, Corporate Exhibition Booth and Poster Presentation
- ・6th Annual Meeting of Reserch Society for Cancer 3-D culture: January 28, 2025, Tokyo, Poster Presentation
- ・29th Annual Meeting of the Japanese Association for Molecular Target Therapy of Cancer: June 18–21, 2024, Tokyo, Poster Presentation: “Comparative Sensitivity Analysis of Antibody-Based Drugs and Small Molecule Drugs Using Cancer Spheroids”
- ・27th Annual Meeting of the Japanese Association for Molecular Target Therapy of Cancer:June 21–23, 2023, Saga, Poster Presentation: “Toward Early Social Implementation of Personalized Cancer Medicine: Drug Sensitivity Testing Using Cancer Stem Cell Spheroids”
Exhibitions
- ・42nd Medicinal Chemistry Symposium: November 18–20, 2025, Tokushima (Awagin Hall), Booth Exhibition
- ・2025Japan Kyoto Trade Exhibition in Dubai:2025/2/10~13、UAE Dubai
- ・110th Annual Meeting of the Japanese Society of Gastroenterology: May 9–11, 2024, Tokushima
- ・114th Annual Meeting of the Pharmaceutical Society of Japan: March 28–31, 2024, Yokohama
Grants and Funding
- ・JST (Japan Science and Technology Agency) START Program (Founded in November 2016 through this program)
- ・Kyoto University Incubation Program
- ・Kansai Mirai Bank: Kansai Mirai Joint Research Grant Program
- ・Kyoto Industry and Technology Advancement Center (Kyoto Sangyo 21): FY2022 SME Collaborative Manufacturing Support Program
- ・Ministry of Economy, Trade and Industry (METI), Kinki Bureau of Economy, Trade and Industry: FY2022 Supplementary Budget Grant for Promotion of Regional Emerging Industry Development (Regional DX Promotion Environment Development Program (Industry-Specific DX Promotion Program)) — Regional Digital Innovation Demonstration Type
- ・Ministry of Economy, Trade and Industry (METI), Kinki Bureau of Economy, Trade and Industry: FY2022 Supplementary Budget Regional DX Promotion Environment Development Program (Industry-Specific DX Promotion Program) — Regional Digital Innovation Demonstration Type
- ・Japan Agency for Medical Research and Development (AMED): FY2022 Innovative Medical Technology Research and Development Promotion Program (Industry-Academia-Government Collaborative Type)
Theme: “Development of Innovative Personalized Medicine Based on Patient-Derived Cancer Stem Cell Culture” - ・Kyoto Industry and Technology Advancement Center (Kyoto Sangyo 21): FY2024 Co-creation Manufacturing Support Program
Title: “Building a Business Implementation Framework for Next-Generation Medical and Drug Discovery Research Systems” - ・Kyoto Prefecture: FY2024 Genki-jirushi (Thriving) SME Certification Program — Certified
Tokyo Future Style is an authorized distributor of Kyo Diagnostics Co., Ltd.
We provide contract manufacturing services through trusted partner manufacturers, with all inquiries and coordination handled through our single point of contact.
- ・One-stop support from reagent procurement to manufacturing requests
- ・Pre-consultation support for technical specifications and requirements clarification
- ・Delivery schedule management and troubleshooting coordination handled by our team
Please contact us for any inquiries, questions, or information requests.
Tokyo Future Style, Inc.
info@tokyofuturestyle.com
TEL:029-851-9222 FAX:029-851-9220


